Draw An Appropriate Lewis Structure For If5
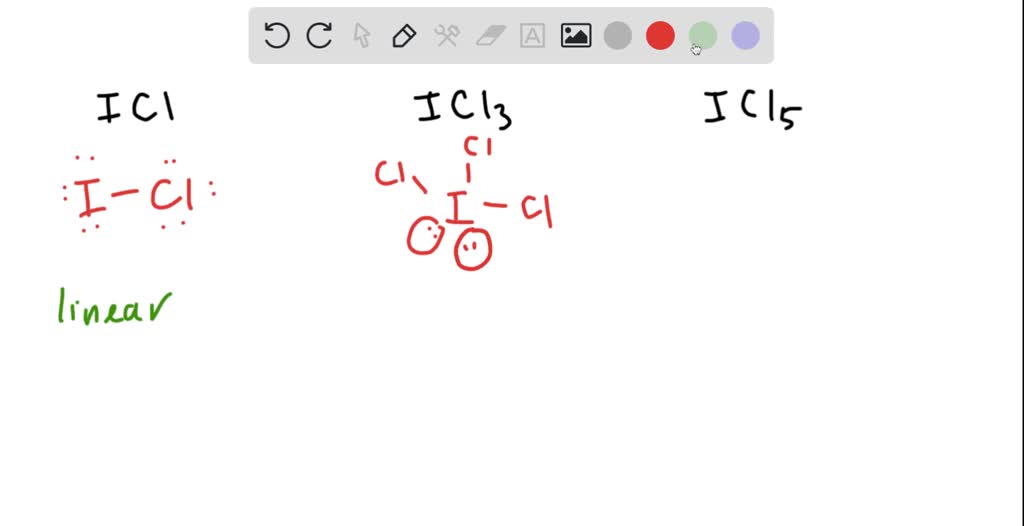
Draw An Appropriate Lewis Structure For If5 - Web 0:00 / 1:40 lewis dot structure of if5 (iodine pentafluoride) kentchemistry.com 24.9k subscribers 48k views 12 years ago every video i quickly take you through how to draw the lewis. For selecting the center atom, you have to remember that the atom which is less. Web the lewis structure (lewis dot diagram) for if5. Web hello, it's time for your daily chemistry dose! Iodine is the least electronegative.
There are a total of 42 valence electrons. Web 5 steps to draw the lewis structure of if5 step #1: Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Web 1) draw a lewis structure for iof5 and calculate the formal charges on each atom. Part a draw an appropriate lewis structure for ifs. Web a video explanation of how to draw the lewis dot structure for iodine pentafluoride, along with information about the compound including formal charges, pola. For if5, we have a total of 42 valence electrons.
IF5 Lewis, VSEPR and Box diagram for hybridiaztion YouTube
Lewis structure of if5 for counting valence electrons around the terminal fluorine atoms. See the big list of lewis structures. Web the lewis structure (lewis dot diagram) for if5. For the if5 lewis structure, calculate the total number of valence electrons for the if5 molecule. 2) draw a lewis structure for if5 and calculate the.
If5 Lewis Structure
Include all lone q (5 ଡ bo identify the geometry of if5 using vsepr theory. Valance electron determination considering the if5 lewis structure, both iodine and fluorine atom contain 7 valence electrons. While selecting the atom, always put the least electronegative atom at the center. Web chemistry questions and answers. Web steps by using the.
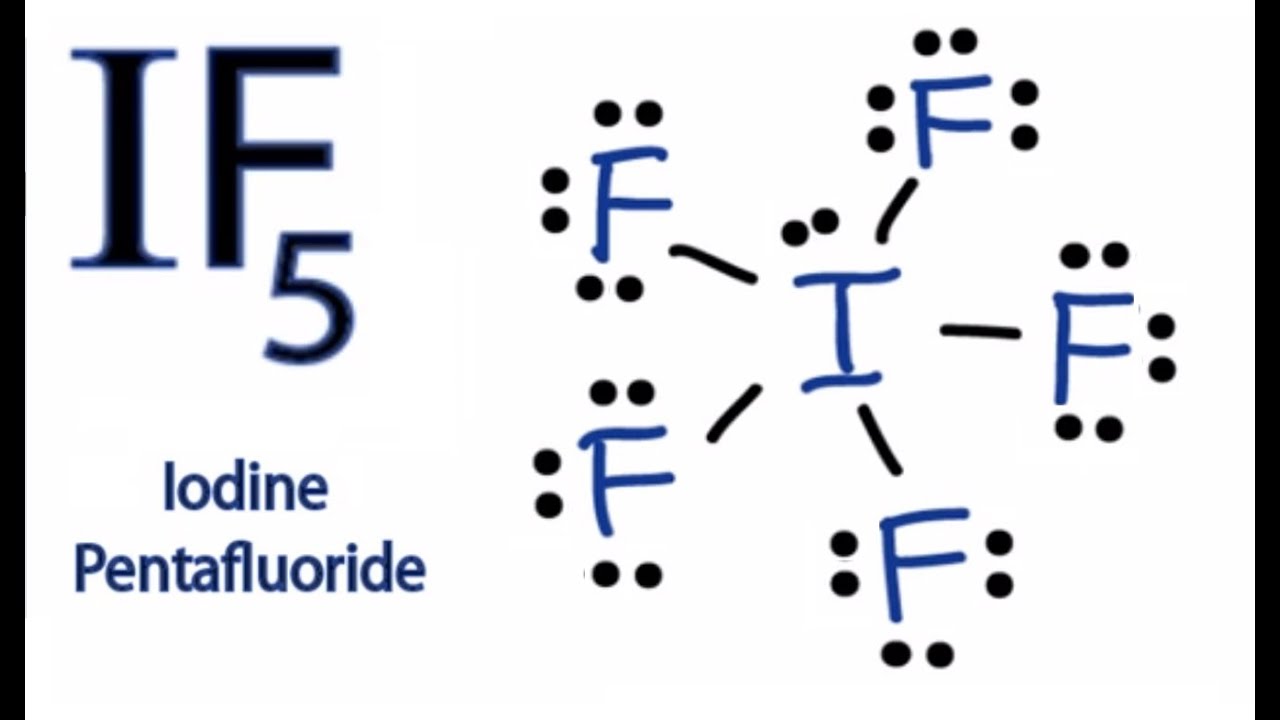
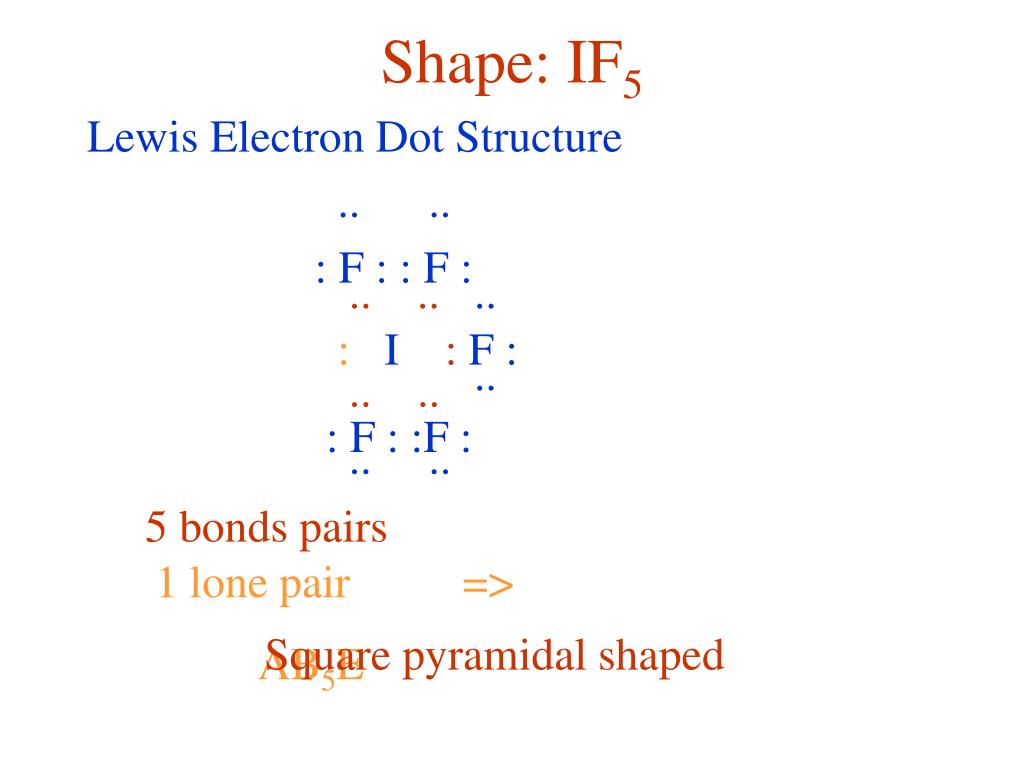
Draw the Lewis structure of iodine pentafluoride, IF5.
Web this widget gets the lewis structure of chemical compounds. The central iodine atom a. For the central iodine atom: Include all lone q (5 ଡ bo identify the geometry of if5 using vsepr theory. Put least electronegative atom in centre 3. For each compound draw an appropriate lewis structure, determine the molecular geometry using.
If5 Lewis Structure
Valance electron determination considering the if5 lewis structure, both iodine and fluorine atom contain 7 valence electrons. Here, the given molecule is if5 (iodine pentafluoride). Web 10k views 3 years ago lewis structures. Web chemistry questions and answers. Figure out how many electrons the molecule must have, based on the number of valence electrons in.
IF5 Lewis Structure How to Draw the Lewis Structure for IF5 YouTube
Web 0:00 / 1:40 lewis dot structure of if5 (iodine pentafluoride) kentchemistry.com 24.9k subscribers 48k views 12 years ago every video i quickly take you through how to draw the lewis. In the case of if5, the iodine atom has 7. Web 1) draw a lewis structure for iof5 and calculate the formal charges on.
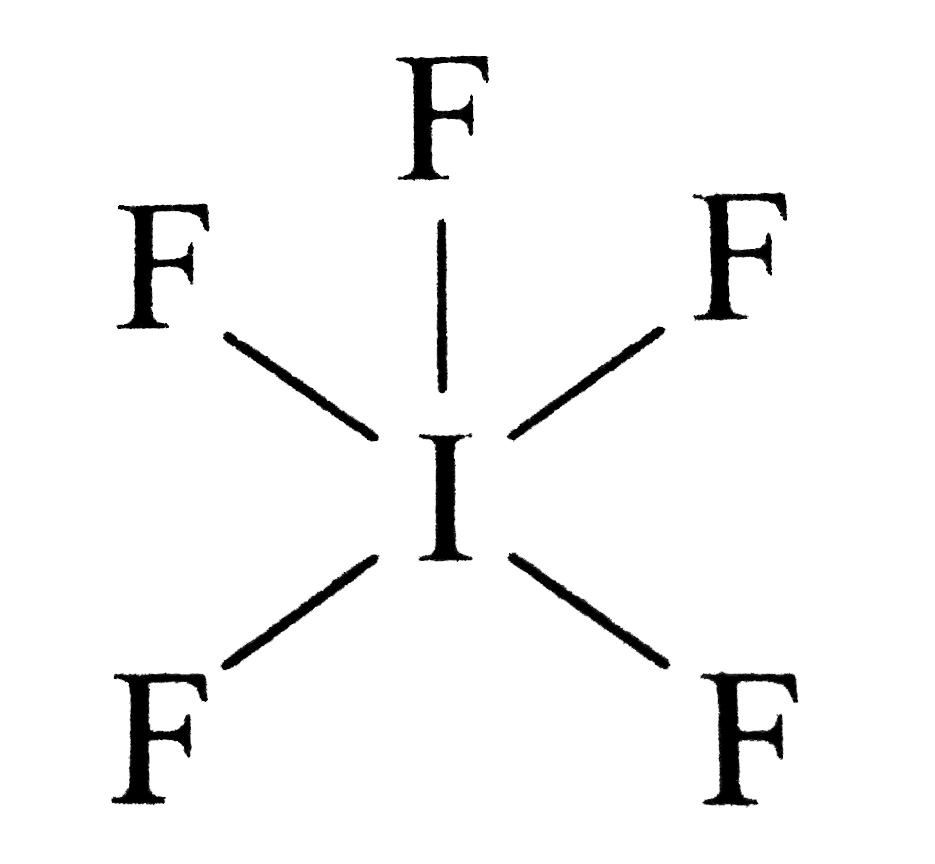
IF5 Molecular Geometry Science Education and Tutorials
This gives it ax5e1 shape by vsepr, and. Remember that iodine (i) can hold more than eight valence electrons. Iodine is the least electronegative. Web in the lewis structure for if5 you'll need to put a total of 12 valence electrons on the iodine atom in order to draw the lewis structure. Web drawing the.
If5 Lewis Structure
Let us follow a few steps. Lewis structure of if5 for counting valence electrons around the terminal fluorine atoms. Web drawing the lewis structure for if 5. Valance electron determination considering the if5 lewis structure, both iodine and fluorine atom contain 7 valence electrons. For the if5 lewis structure, calculate the total number of valence.
If5 Lewis Structure
Iodine pentafluoride (if5) has 5 fluorine atoms bonded to a central iodine, and that iodine has one lone pair on it. The number of lone pairs = the number of single bonds = the number of double bonds = 2. While selecting the atom, always put the least electronegative atom at the center. Web drawing.
IF5 Molecular Geometry, Bond Angles and Electron Geometry YouTube
For selecting the center atom, you have to remember that the atom which is less. Web in the lewis structure for if5 you'll need to put a total of 12 valence electrons on the iodine atom in order to draw the lewis structure. Web 5 steps to draw the lewis structure of if5 step #1:.
If5 Lewis Structure
Iodine is below period two on the periodic table so it can have. For if5, we have a total of 42 valence electrons. See the big list of lewis structures. Web drawing the lewis structure for if 5. Put least electronegative atom in centre 3. Put one electron pair in each bond 4. #1 draw.
Draw An Appropriate Lewis Structure For If5 Here, the given molecule is if5 (iodine pentafluoride). Put one electron pair in each bond 4. Find more chemistry widgets in wolfram|alpha. Find the total valence electrons in if5 molecule. In the case of if5, the iodine atom has 7.
Remember That Iodine (I) Can Hold More Than Eight Valence Electrons.
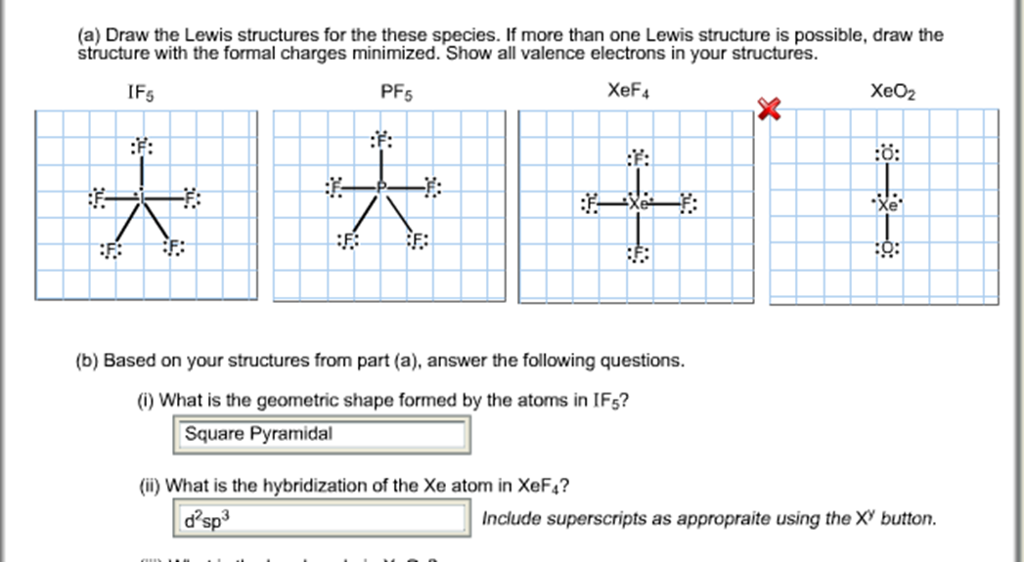
Web in the lewis structure for if5 you'll need to put a total of 12 valence electrons on the iodine atom in order to draw the lewis structure. Determine the total number of valence (outer shell) electrons. For each compound draw an appropriate lewis structure, determine the molecular geometry using vsepr theory, determine whether the molecule is polar and identify the hybridization of all interior atoms: For the central iodine atom:
Web 1) Draw A Lewis Structure For Iof5 And Calculate The Formal Charges On Each Atom.
Include all lone q (5 ଡ bo identify the geometry of if5 using vsepr theory. Lewis structure of if5 for counting valence electrons around the terminal fluorine atoms. The sum of the valence electrons is 5 (from n) + 6 (from o) = 11. Web steps to properly draw the if 5 lewis structure, follow these steps:
Valance Electron Determination Considering The If5 Lewis Structure, Both Iodine And Fluorine Atom Contain 7 Valence Electrons.
In the case of if5, the iodine atom has 7. Web drawing lewis structures for molecules with one central atom: Part a draw an appropriate lewis structure for ifs. Web 5 steps to draw the lewis structure of if5 step #1:
For Selecting The Center Atom, You Have To Remember That The Atom Which Is Less.
In order to find the total valence electrons in a if5 (iodine. Web steps by using the following steps, you can easily draw the lewis structure of if 5: Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Find more chemistry widgets in wolfram|alpha.