Draw The Electron Configuration For A Neutral Atom Of Nitrogen
Draw The Electron Configuration For A Neutral Atom Of Nitrogen - At oxygen, with z = 8 and eight electrons, we have no choice. The number of shells shows which period, or row, it’s in and the number of electrons in the outer shell shows which group it’s in. Nitrogen (atomic number 7) fills the 1s and 2s subshells and has one electron in each of the three 2p orbitals, in accordance with hund’s rule. Electron configuration of fluorine (f) [he] 2s 2 2p 5: This problem has been solved!
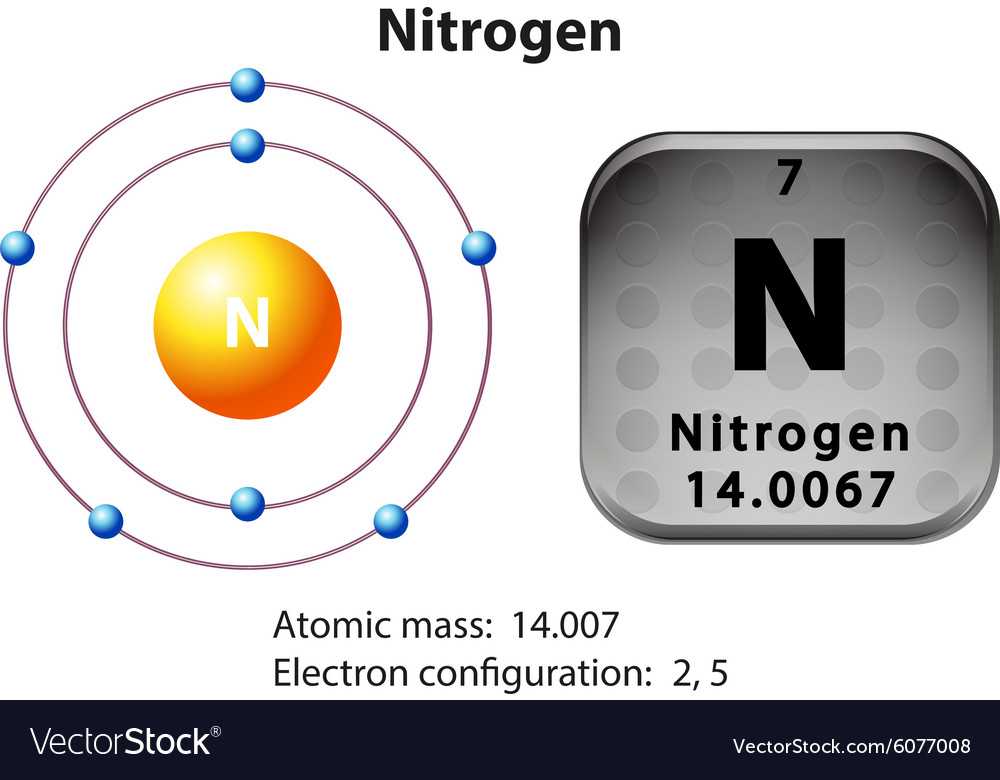
Web if we look at the correct electron configuration of the nitrogen (z = 7) atom, a very important element in the biology of plants: How many valance electrons are there in the ground state electron configuration of a neutral phosphorus atom. Electron configuration of neon (ne) [he] 2s 2 2p 6: Web draw the electron configuration for a neutral atom of nitrogen. How to write electron configurations. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Electron configuration of oxygen (o) [he] 2s 2 2p 4:
Nitrogen Electron Configuration (N) with Orbital Diagram
A neutral helium atom, with an atomic number of. Electron configuration can be done in two ways. Web for hydrogen, therefore, the single electron is placed in the 1 s orbital, which is the orbital lowest in energy (figure 6.8.1 6.8. Based on hund's rule , one electron fills each p \rm p p orbital,.
Orbital Diagram For Nitrogen (N) Nitrogen Electron Configuration
Energy 1 х this problem has been solved! An atom has a valence shell electron configuration of #ns^1#. The number of shells shows which period, or row, it’s in and the number of electrons in the outer shell shows which group it’s in. This is the number of protons in the nuclei of nitrogen atoms..
How to write the Electronic Configuration of Nitrogen Chemical
At oxygen, with z = 8 and eight electrons, we have no choice. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw a lewis structure, showing all bonding and nonbonding electrons, for the oxygen molecule and for the nitrogen molecule. The element atomic number and name are.
Nitrogen Table of Elements by Shrenil Sharma
Nitrogen (atomic number 7) fills the 1s and 2s subshells and has one electron in each of the three 2p orbitals, in accordance with hund’s rule. Web all of the electrons in the noble gas neon (atomic number 10) are paired, and all of the orbitals in the n = 1 and the n =.
Nitrogen Facts, Symbol, Discovery, Properties, Uses
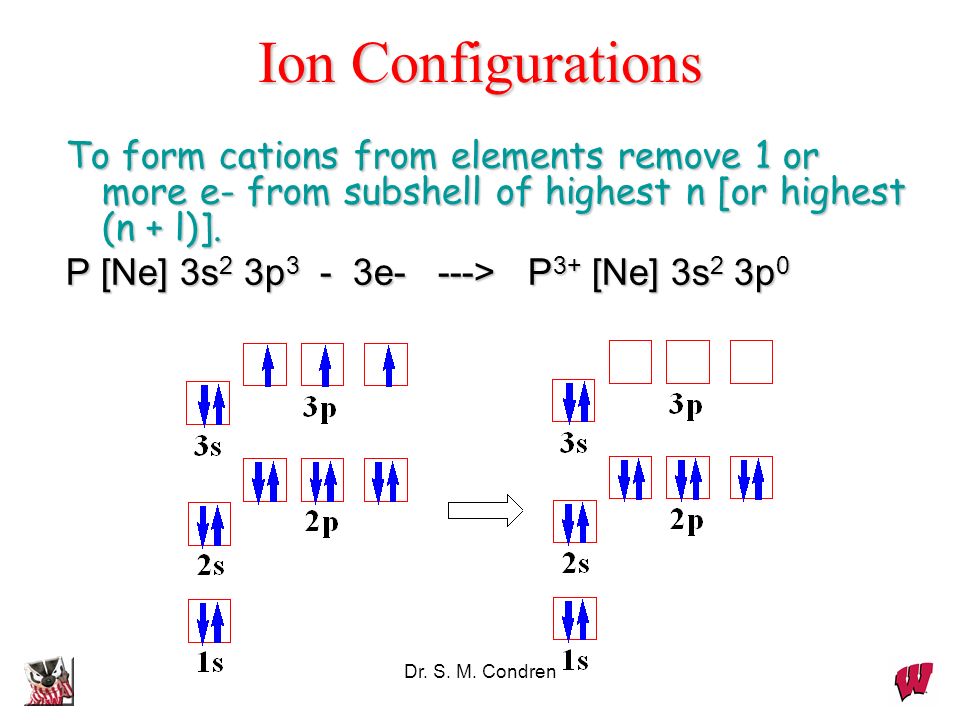
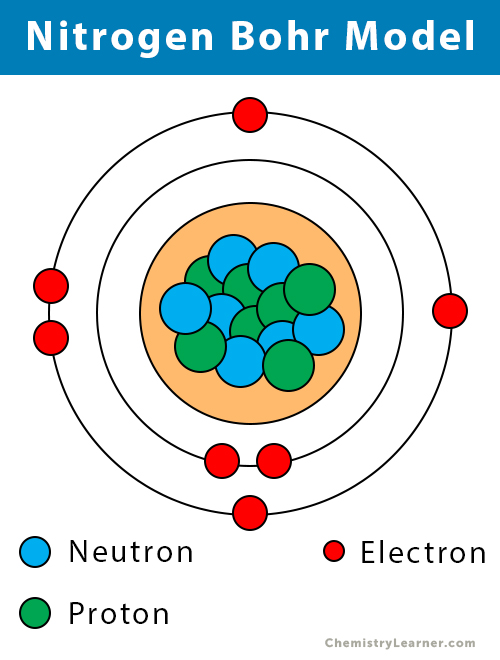
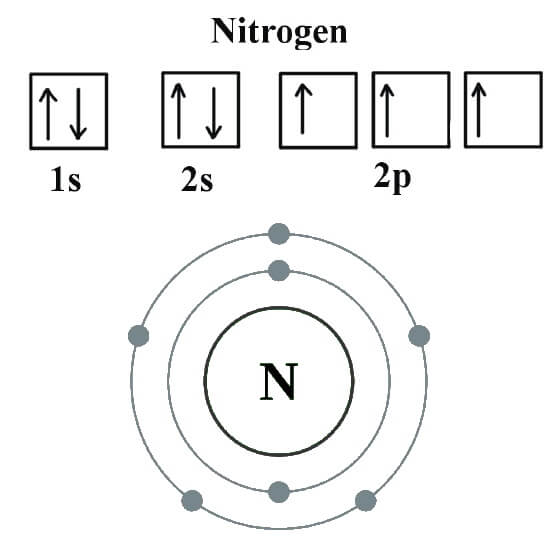
The atomic number of nitrogen is 7. Web the electron configuration of nitrogen is thus 1s 2 2s 2 2p 3. This is the number of protons in the nuclei of nitrogen atoms. Web an electrically neutral atom has the following electron configuration: Based on hund's rule , one electron fills each p \rm p.
Electron Configuration Chemistry LibreTexts
The element atomic number and name are listed in the upper left. Web an electrically neutral atom has the following electron configuration: At oxygen, with z = 8 and. Web the arrangement of electrons in nitrogen in specific rules in different orbits and orbitals is called the electron configuration of nitrogen. 1s 2 2s 2.
Symbol and electron diagram for nitrogen Vector Image
An atom has a valence shell electron configuration of #ns^1#. The atomic number of nitrogen is 7. The number of shells shows which period, or row, it’s in and the number of electrons in the outer shell shows which group it’s in. You'll get a detailed solution from a subject matter expert that helps you.
Nitrogen Element With Reaction, Properties, Uses, & Price Periodic Table
Web chemistry chemistry questions and answers write the electron configuration for a neutral atom of nitrogen. Electron configuration of fluorine (f) [he] 2s 2 2p 5: Web the arrangement of electrons in nitrogen in specific rules in different orbits and orbitals is called the electron configuration of nitrogen. Based on hund's rule , one electron.
How many valence electrons does nitrogen have? Ask4Essay
Nitrogen (atomic number 7) fills the 1s and 2s subshells and has one electron in each of the three 2p orbitals, in accordance with hund’s rule. The alkali metal sodium (atomic number 11) has one more electron than the neon atom. Neutral atom of nitrogen will have equal number of proton and electron i.e equal.
Diagram representation of the element nitrogen Vector Image
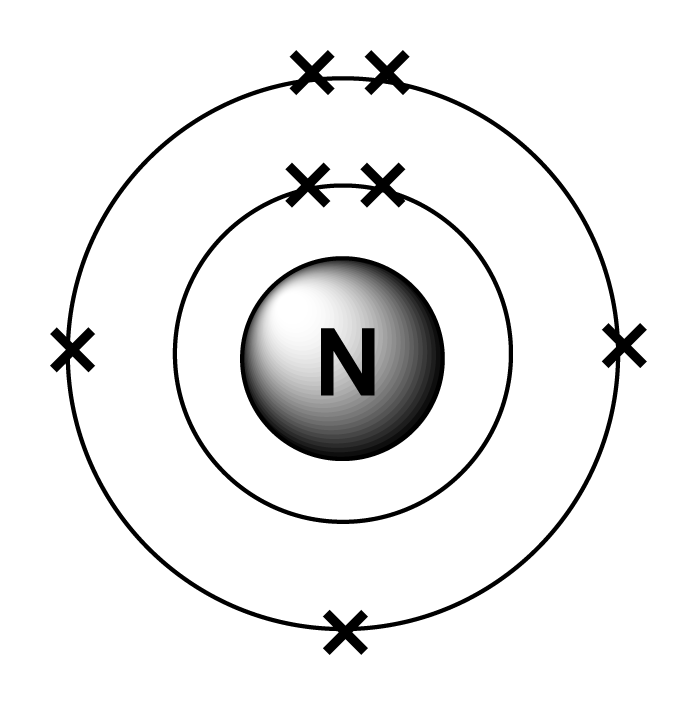
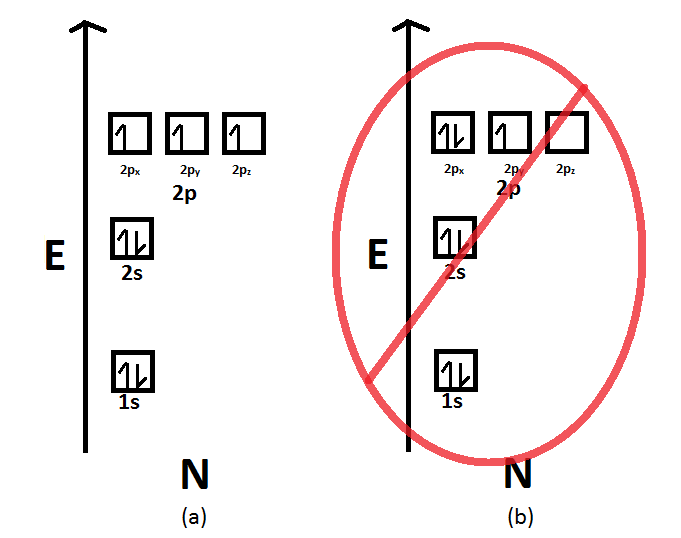
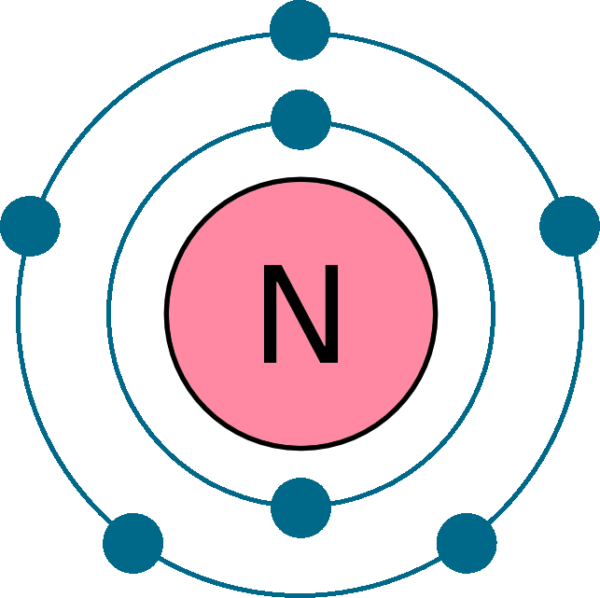
The electron shells are shown, moving outward from the nucleus. Web draw an orbital diagram for nitrogen, z = 7. Electron configuration of fluorine (f) [he] 2s 2 2p 5: The element atomic number and name are listed in the upper left. Web the electron configuration and orbital diagram for carbon are: Web the arrangement.
Draw The Electron Configuration For A Neutral Atom Of Nitrogen A neutral atom has the same number of electrons as protons. 1s 2 2s 2 2p 5: So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). Therefore, its ground state electronic configuration can be written as 1s 2 2s 2 2p 6 3s 2 3p 5. A neutral helium atom, with an atomic number of.
The Arrangement Of An Element’s Electrons Tells You Where It Is On The Periodic Table.
Web the electron configuration of nitrogen is thus 1s 2 2s 2 2p 3. Web chemistry chemistry questions and answers write the electron configuration for a neutral atom of nitrogen. The electron shells are shown, moving outward from the nucleus. These three electrons have unpaired spins.
#1S^2, 2 S^2, 2P^6, 3S^2, 3P^4#.
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web the arrangement of electrons in nitrogen in specific rules in different orbits and orbitals is called the electron configuration of nitrogen. Web the neutral atom chlorine (z=17), for instance has 17 electrons. Electron configuration of oxygen (o) [he] 2s 2 2p 4:
1S 2 2S 2 2P 5:
Web if we look at the correct electron configuration of the nitrogen (z = 7) atom, a very important element in the biology of plants: The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Web draw the electron configuration for a neutral atom of nitrogen. Write the electron configuration for a neutral atom of nitrogen.
This Is The Number Of Protons In The Nuclei Of Nitrogen Atoms.
Web draw an orbital diagram for nitrogen, z = 7. 1s 2 2s 2 2p 6: Web the electron configuration and orbital diagram for carbon are: At oxygen, with z = 8 and.