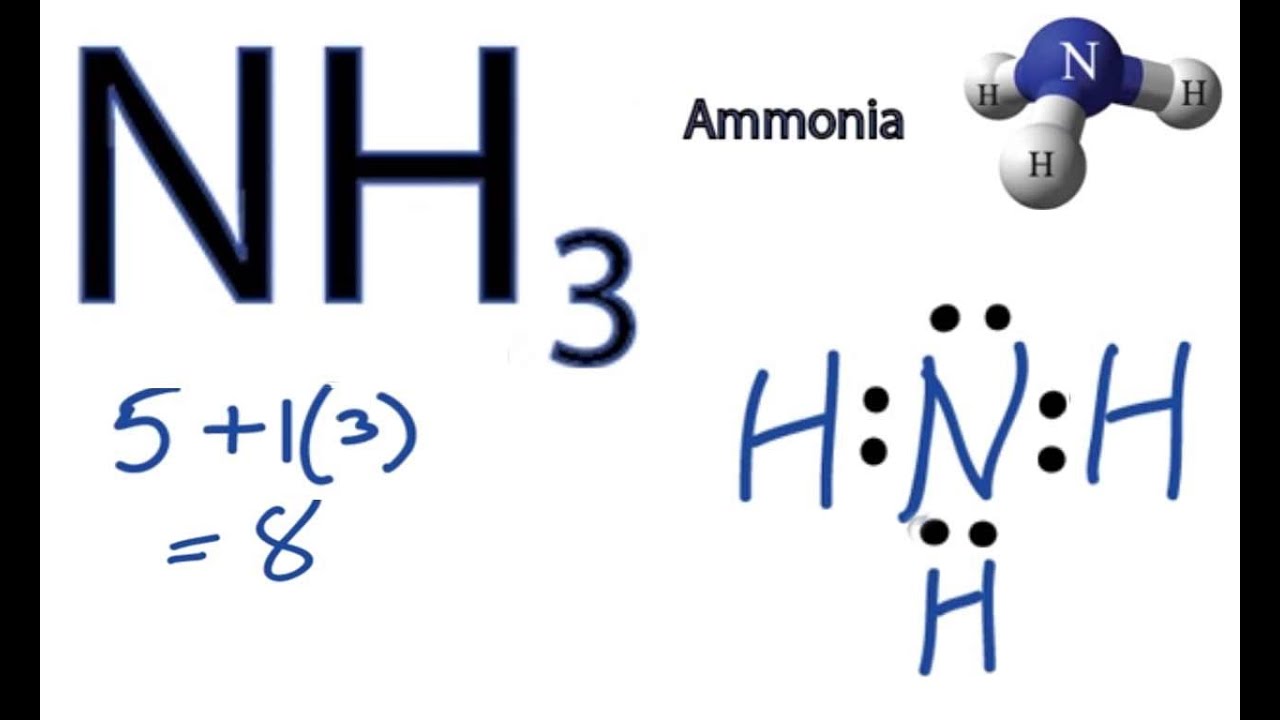Draw The Lewis Structure Of Ammonia Nh3
Draw The Lewis Structure Of Ammonia Nh3 - In this article, we will explore the nh3 lewis structure, also known as ammonia. It is important to accurately distribute the valence electrons, identify the central atom, calculate formal charges, and determine bond pairs and lone pairs. This problem has been solved! Determine the total number of valence electrons for all the atoms in nh3, which is 5 (for nitrogen) + (3 x 1) (for hydrogen) = 8. For resonance structures there must be a double or triple bond present, which is not the case.
+ this problem has been solved! Place the nitrogen atom in the center of the structure and the three hydrogen atoms around it. Web we draw lewis structures to predict: Number of electron regions in ammonia. Web draw the lewis structure of ammonia (nh3). In this article, we will explore the nh3 lewis structure, also known as ammonia. Place an h under the n.
NH3 Lewis Structure How to Draw the Dot Structure for NH3 YouTube
Now that we know the valence electrons for the molecule, we can predict its lewis structure. Now, write down h, n, and h in a horizontal line. Web steps involved in the nh3 lewis structure: You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Hydrogen atoms never take.
Lewis Structure of NH3 (Ammonia) YouTube
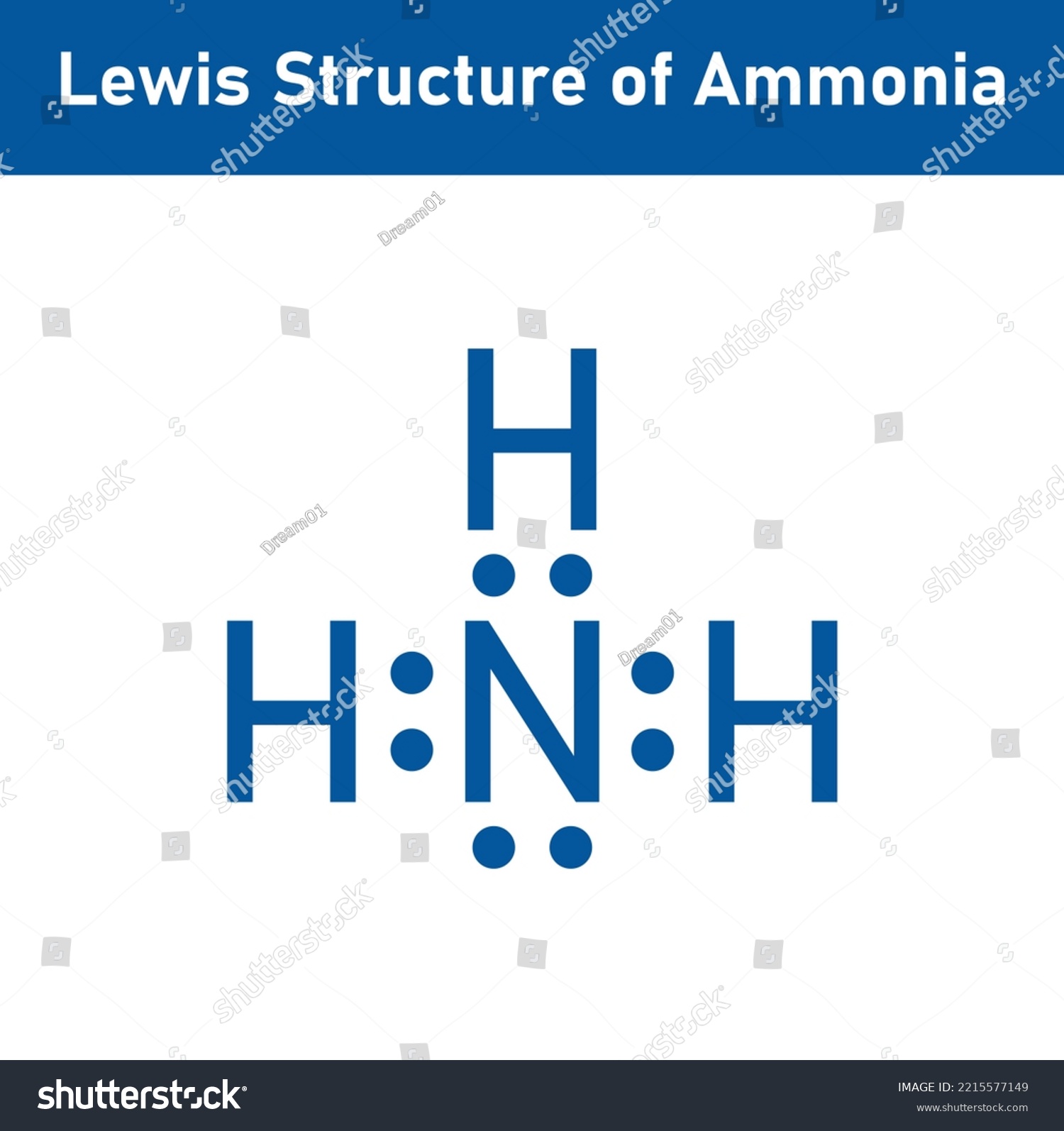
Now, write down h, n, and h in a horizontal line. Web to summarize, the lewis structure of nh3 (ammonia) can be drawn by following the steps mentioned above. How many bonding pairs are there around the n atom? This problem has been solved! Web the lewis structure of the tetra atomic ammonia (nh3) molecule.
Ammonia NH3 Molecule. Skeletal Formula Stock Vector Illustration of
Web to summarize, the lewis structure of nh3 (ammonia) can be drawn by following the steps mentioned above. Now, write down h, n, and h in a horizontal line. It is important to accurately distribute the valence electrons, identify the central atom, calculate formal charges, and determine bond pairs and lone pairs. Web draw the.
How to draw NH3 Lewis Structure? Science Education and Tutorials
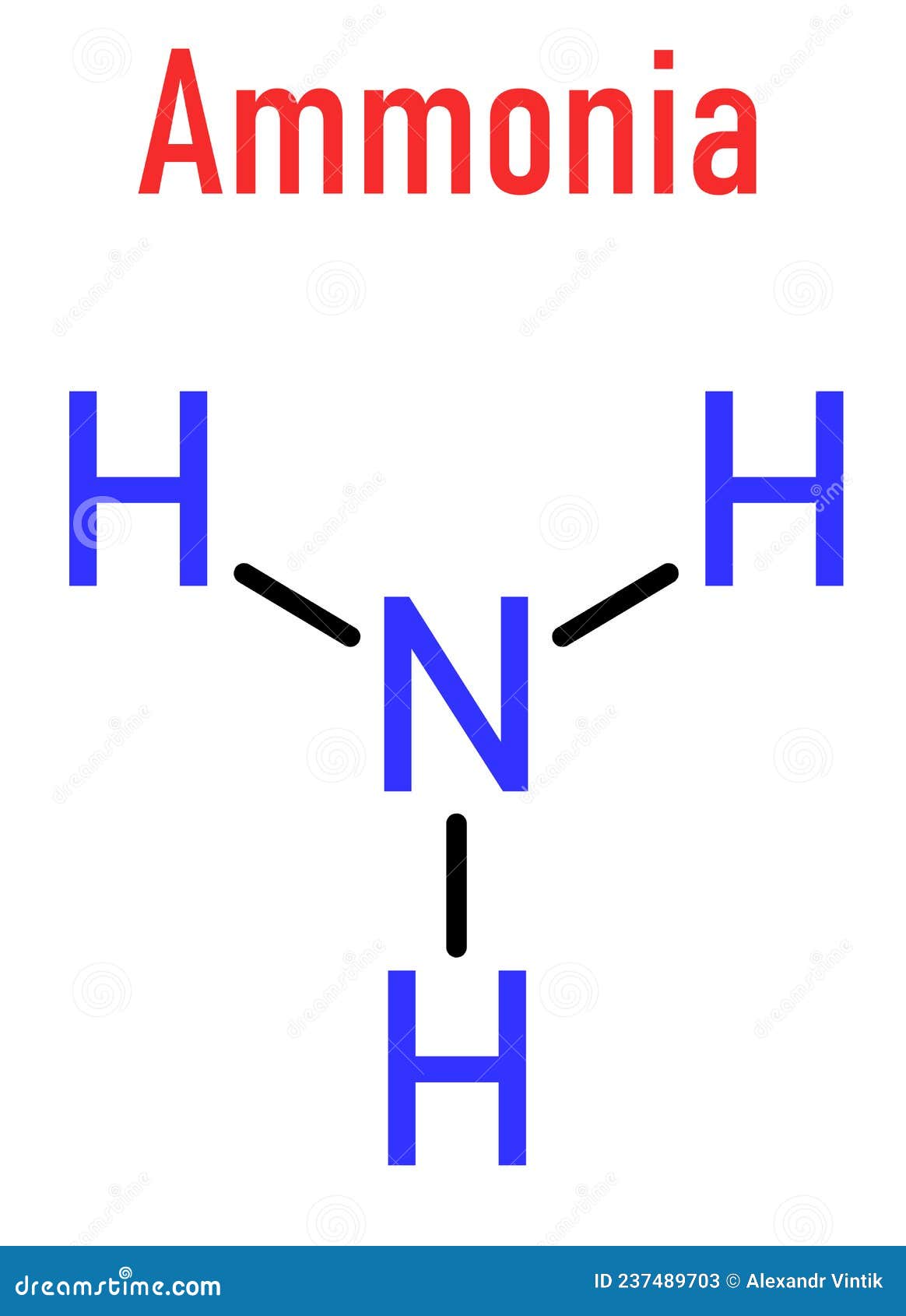
Understanding the molecular structure of ammonia The formula for ammonia is nh3. Draw a single bond between each hydrogen atom and the nitrogen atom. Web the lewis structure of the tetra atomic ammonia (nh3) molecule has three single sigma bonds between the nitrogen and the hydrogen atoms. Count total valence electrons in nh3 first of.
NH3 (ammonia) Lewis dot structure YouTube
The formula for ammonia is nh3. Web steps for drawing the lewis dot structure for nh3 1. This is the reason why ammonia acts as a lewis base, as it can donate those electrons. Place an h under the n. In this article, we will explore the nh3 lewis structure, also known as ammonia. Web.
Lewis Dot Diagram Of Nh3
Web welcome to warren institute! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. This is the reason why ammonia acts as a lewis base, as it can donate those electrons. This chemistry video tutorial explains how to draw the lewis structure of nh3 also known as ammonia..
Draw the Lewis Structure for the Conjugate Acid of Ammonia En
Also place two dots above the n. Moreover, the presence of a single lone pair of electrons on the nitrogen atom is responsible for the bent geometrical structure of the nh3 molecule. Web science chemistry chemistry questions and answers draw a lewis structure for ammonia (nh3). Web 6 steps to draw the lewis structure of.
Nh3 ammonia molecule Royalty Free Vector Image
There is really only one way to draw the lewis structure for ammonia (nh3). Draw a lewis structure for ammonia (nh3). For resonance structures there must be a double or triple bond present, which is not the case. Determine the total number of valence electrons for all the atoms in nh3, which is 5 (for.
Estructura de Lewis NH3, Amoniaco » Quimica Online
Here, the given molecule is nh3 (ammonia). Moreover, the presence of a single lone pair of electrons on the nitrogen atom is responsible for the bent geometrical structure of the nh3 molecule. For the nh3 structure use the periodic table to find the total number of valence electrons for the. Place two dots in between.
Lewis Structure Ammonia Nh3 Scientific Vector Stock Vector (Royalty
Moreover, the presence of a single lone pair of electrons on the nitrogen atom is responsible for the bent geometrical structure of the nh3 molecule. This chemistry video tutorial explains how to draw the lewis structure of nh3 also known as ammonia. It is important to accurately distribute the valence electrons, identify the central atom,.
Draw The Lewis Structure Of Ammonia Nh3 In order to draw the lewis structure of nh3, first of all you have to find the total number of valence electrons present in the nh3 molecule. Hydrogen atoms never take the central position, so we will place the nitrogen atom in the center. How many bonding pairs are there around the n atom? Here, the given molecule is nh3 (ammonia). Web learn more understanding the nh3 lewis structure is crucial for comprehending the chemical properties and behavior of ammonia.
Draw A Single Bond Between Each Hydrogen Atom And The Nitrogen Atom.
Total electron region is taken by the summation of sigma bonds and lone pairs around relevant atom. + this problem has been solved! Web in this video, you will learn how to draw the lewis structure for chemicals based on total valence electrons, the octet rule, duet rule, and also you will learn about lone pair and bonding pair. Web 6 steps to draw the lewis structure of nh3 step #1:
Place An H Under The N.
Now that we know the valence electrons for the molecule, we can predict its lewis structure. Web welcome to warren institute! To decide the geometry, shape and hybridization of a molecule, drawing the correct lewis structure is very important. Web steps involved in the nh3 lewis structure:
Web The Lewis Structure Of The Tetra Atomic Ammonia (Nh3) Molecule Has Three Single Sigma Bonds Between The Nitrogen And The Hydrogen Atoms.
Find the total valence electrons in nh3 molecule in order to find the total valence electrons in nh3 molecule , first of all you should know the valence electrons present in nitrogen atom as. Web we draw lewis structures to predict: This problem has been solved! Drawing the lewis structure for nh 3 ( ammmonia)
Hydrogen Atoms Never Take The Central Position, So We Will Place The Nitrogen Atom In The Center.
In order to draw the lewis structure of nh3, first of all you have to find the total number of valence electrons present in the nh3 molecule. Place the nitrogen atom in the center of the structure and the three hydrogen atoms around it. Now, write down h, n, and h in a horizontal line. Number of electron regions around nitrogen atom in ammonia